Fields of research
Current research projects
Elastic properties of muscles
During natural movements, muscles exhibit a variety of functions; they serve not only as motors, but also as springs, brakes, and struts. Our lab seeks to understand the mechanisms underlying the spring properties exhibited by muscles during active lengthening and shortening.
Molecular titin
Additional information on the Winding Filament Hypothesis and titin’s role.
Molecular approach: Exploring the Winding Filament Hypothesis through Transmission Electron Microscopy
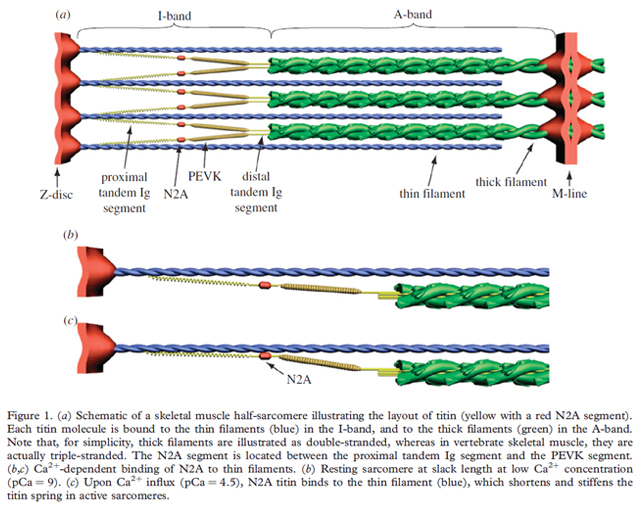
Over the last several years, our understanding of titin’s contributions to passive and active muscle properties has exploded. Several models and theories have grown out of this work, most notably the winding filament hypothesis (WFH). This model fills existing muscle theory gaps while building on the sliding filament theory. In the WFHs, the N2A region of titin binds to actin upon Ca2+ influx. The PEVK segment of titin (which lies next to the N2A region) winds on the thin filaments during force development because the cross-bridges not only translate but also rotate the thin filaments. As a muscle shortens or lengthens, it is theorized that the titn’s components interact in such a way that when the muscle is activated, titin acts as a powerful elastic spring. Titin’s active properties are believed to fill in the gaps unresolved by the Hill muscle model. We are working to demonstrate this model’s feasibility by measuring the passive and active “stretches” of titin’s elastic elements, PEVK and tandon Ig domain regions. The winding filament hypothesis predicts that both the proximal tandem Ig and PEVK domains extend in sarcomeres from passively stretched muscles, but only PEVK will extend in actively stretched muscles due to N2A’s interaction with the thin filament. Previous work on a N2A deficient mouse model (mdm mice) have, thus far, supported the WFH.
The present research will determine the location of the N2A epitope within the I-band and estimate force-length relationships for the proximal tandem Ig and PEVK segments in passive and activated soleus muscles from wild type and mdm mice. We believe that in mdm muscles, the proximal tandem Ig and PEVK domains should extend in sarcomeres from both passive and activated muscles. Wild-type and mdm muscles will differ in force-extension behavior of the proximal tandem Ig and PEVK domains of titin. Passive and activated muscles will differ in force-extension behavior of the proximal tandem Ig and PEVK domains.
For this work, antibody-labeling studies are critical for understanding contributions of titin to active muscle force. Following in the footsteps of previous work, we will use immunogold labeling with a polyclonal titin N2A antibody to locate the N2A epitope within the I-band and to estimate the force-length relationships of the proximal tandem Ig and PEVK segments.
Prepared muscles, fixed at a range of lengths and forces from wild-type and mdm mice will be embed in a porous plastic resin (LR White), sectioned at 40 nm parallel to the muscle fiber orientation and treated with a polyclonal N2A primary antibody and secondary antibody. This antibody is specific for the primary antibody and has been attached to a colloidal gold particle. Gold particles are visible on transmission electron micrographs, labeling the N2A region. This will allow us to calculate the distances between the antibody label, the edge of the A-band, and the center of the Z-line. From these data, the location of the N2A epitope, and force-extension curves for the proximal tandem Ig and PEVK regions of titin will be obtained. To date, we have developed and implemented a protocol for passive muscle. Active muscle protocols have been developed and are in their testing stages. The implications of this work, will solidify the WFH into our understanding of the complex properties of muscle contractions.
Sports Science–research by Anthony Hessel
The applications of residual force enhancement: Eccentric Contraction Training
Women are 2-8 times more likely to suffer a knee injury than men who are similarly active. Current training approaches do not do enough to lower a woman’s risk of knee injury. We are trying to improve on preventative workouts that protect the knee from injury.
A fundamental aspect of my research is to use what we learn about muscles to develop effective training strategies for a wide variety of situations. As a former NCAA athlete and current triathlete, I have a generalized interest in sports science. Lately, my focus has been on preventative strategies for non-contact knee joint injuries (ACL tears, knee strains). My muscle research is focused on the molecular mechanism of eccentric contractions. I am very interested in these lengthening contractions (muscle that are flexing while getting longer) and how we can wield these contractions during workouts to prevent injury, increase successful ageing and expedite recovery from reconstructive surgery.
With over 150,000 knee injuries occurring from non-contact athletic events, preventative strategies are warranted to limit knee injury risk. In athletic competitions, knee injuries are most commonly associated with a quick change of direction, or speed change (i.e., cutting in basketball, football or soccer). These cutting events put an incredible amount of force on the knee, with femur and tibial torques rotating the knee joint in opposite directions. Biomechanics studies that focus on the knee-abduction moment (KAM), an index of knee stability, report that the higher the KAM value, the greater the risk of knee injury. Several factors influence KAM values; the most important are knee valgus moment, body mass, and quadriceps muscle strength. In women, KAM values are physiologically higher than in men, at least partially explaining the higher incidence of injury or re-injury in women. Women are 2-8 times more likely
to sustain a knee injury then men due to a fundamentally destabilized knee caused by relatively weak muscles and ligaments (even in professional athletes).
Current exercise strategies to increase knee stability focus on agility and strength. Cutting motion torque forces that are applied to the knee joint are usually countered by the muscles that surround the knee (i.e. quadriceps) as a first line of defense. Once the external torques on the knee overpower the leg muscles, the stress is placed on the knee ligaments of the knee joint (ACL, LCL, PCL, MCL), which under enough strain, will tear. Ligaments are slow to heal when strained (due to low blood flow), while torn ligaments never heal. To protect the ligaments from strain, most preventative training strategies focus on strengthening the quadriceps that surround the knee, so that they can counter more of the external knee torque forces. Usually, simple exercises like lunges or squats (weighted or not) are used for this purpose. Agility practice (i.e. rope ladder steps) is also used to build the athlete’s balance and posture, which
minimizes external knee torques by proper locomotion form. The last decade of research has seen different approaches developed to target knee strength and increase knee stability.
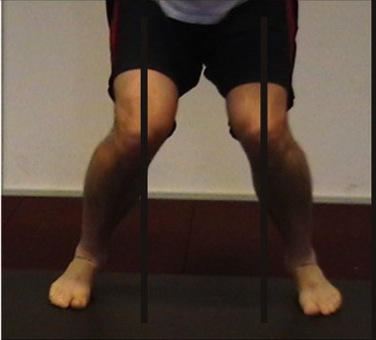
Valgus is commonly called knock-knee, it is caused by a rotation of the tibia and femur due to a lack on proper leg strength. There is also varus, which is commonly called bow-legged, which has the opposite knee rotation. Most of us get both valgus and varus after a long lunge or squat session, when our legs get “wobbly”.
During many typical cutting motions, the leg muscles are over powered by the force being exerted on the leg to the direction pivot. This places the athlete in to a valgus, which if server enough can lead to ligament damage, require surgery to repair.
Most researchers agree that both strength and agility exercises are a good preventative strategy, but the convention, just described, for attaining these goals are time consuming, inefficient and not aggressive enough. Further, since both strength and agility are important, implementing a single activity that increases strength and agility quickly would place a limited strain on precious practice time.
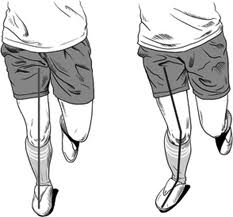
Eccentric overloading workouts (EOW) have shown great potential over the past decade as a muscle, tendon, and ligament enhancer. Eccentric muscle contractions are active muscles that are lengthening (consider a bicep curl, when you lower the weight, that is eccentric contractions). Eccentric contractions can produce more force than concentric contractions (picking up the weight during a bicep curl), allowing greater loads to be placed on a muscle system during eccentric stretch, which further stimulates the muscular mechanisms that build muscle and tendon structures. In fact, only eccentric style workouts have been shown to restructure tendon systems successfully. EOW are unique, in that they maximize eccentrics characteristics to place very large loads on muscles, which rapidly build muscle/tendon systems, as well as proprioceptive mechanisms needed for balance and agility. Furthermore, when compared to conventional practices, EOW are successful with very short bouts once or twice a week. Due to these attributes, personal trainers and coaches have been incorporating them into their workout routines aggressively. Though these workouts show promise, the primary literature on which style of exercise (conventional squats vs. EOW) are the most effective strategy for knee protection. A common EOW implemented by coaches and personal trainers are box-hop jumps (BHJ). For the BHJ, in short, the athlete jumps off an approx. 2 foot box, lands on both feet, squats and then quickly jumps into the air. When landing, the subject lands flat-footed on one foot with a bent knee and then immediately flat-foot hops ten meters, switches feet and hops back to the start position. The amount of repetitions, height of the box and length of the hops can all be altered as the athlete’s performance improves. This exercise uses the fundamental concept of gravity and the athletes body weight to apply forces 3-8 times their body weight on single leg muscle groups in an eccentric fashion. More importantly, the muscle will have a force applied to them that is 1.5-3 times larger than what would be possible in conventional concentric workouts.
–Athletes and scientists are only starting to appreciate the application of eccentric training. You can lift more eccentrically (ex: lowering a dumbbell) then concentrically (ex lifting up a dumbbell) without placing a large burden on the metabolic or cardiovascular system, which has drastic implications for muscle strength, ligament and tendon restructuring and geriatric medicine.
Current project status:
We are currently using NAU Physical Educational Science Classes (workout classes) to compare how the incorporation of 3-5 minute bout of EOW’s into a normal routine improve knee stability. We have baseline assessments, and the students are in the middle of the training protocol. More to come in May 2015, when the first round of this study ends.
Historical research projects
Neuromechanics of prey capture in frogs
Feeding in frogs consists of coordinated head, jaw, and tongue movements that serve as an excellent model for studying the relationship between biomechanics and control in vertebrates. We have focused on feeding studies in the following anuran genera: Bufo, Rana, Litoria, Hemisus.
Diversity of form, function, and control
While herps represent a dizzying range of forms used in an equally diverse functions, we also enjoy comparing the biomechanics of these animals to the truly astonishing diversity represented by invertebrate organisms. As such, we’ve looked at the muscle mechanical properties of orthopterans and developed a research project looking at structure and function of dynamic compliance in cephalopod vasculature.
Jumping salamanders
Graduate student researcher Anthony Hessel studied lungless salamanders (Plethodontidae), who have a unique jumping mechanism that does not use their legs for propulsion into air. Plethodontid salamanders have been observed to jump using a unique mechanism in which they bend and unbend their torsos laterally with such force that they hurl themselves into the air (Ryerson, 2013). We have attempted to understand this mechanism by performing a detailed biomechanical analysis. So far, we have learned that the salamander does not really bend and unbend laterally. Instead, the salamander bends its body into a C-shape and then pushes that bend into the hips (think of a wave caused by moving a stretched-out slinky from side to side). As the wave moves through the hips, they rotate; this rotation pulls on the ipsilateral (to the bend) hind limb, which is planted in front of the hip. Because the movement of the hip is away from the planted foot and the foot is anchored to the ground, the hip rotates towards the foot at such speed (17 body lengths a second!) that it catapults the salamander over the planted foot, achieving liftoff. (Consider rock climbing: you reach way over your head and grab a ledge. At this point, your shoulders are not lined up because of the arm stretch. If you bring your free hand’s shoulder to the level of the anchored shoulder, you will raise your body. The salamander jump works the same way, except this happens so quickly that it throws the salamander over the planted foot). Due to the importance of hip rotation to the performance of the jump, we call this mechanism “The Hip-Twist Jump.” Check out a video of the jump. There are also some interesting biomimetic applications to consider. Salamander jumping shows us that there are alternative ways to get into the air, even with an object that lies flat on the ground and has no means of “pushing” directly down into the ground. A piece of equipment could be placed covertly in the ground, and when triggered, flung into the air. Studying the salamander has resulted in considering designs for a “flat catapult.” More information on the salamander research.
Listed below are abstracts describing specific research projects undertaken by the lab members:
Spring properties of muscle during active shortening and lengthening
Jenna Monroy, Jim Hokanson, & Kiisa Nishikawa
When its load is reduced, an active muscle will recoil elastically to a shorter length. When its load increases, an active muscle will lengthen. In this study, we describe and compare the spring properties of mouse soleus muscle during active shortening and lengthening. Using load-clamp and dynamic stiffness experiments performed on a servo-motor force-lever, we measured the changes in muscle length that resulted from changes in muscle force. Using these data, we calculated muscle stiffness across a range of muscle forces and lengths. Our results show that muscle stiffness increases with force (i.e., up to maximum isometric force) and is greatest when changes in load are small (i.e., isotonic shortening). The relationship between change in length and force is exponential during active shortening. Thus, muscle stiffness decreases non-linearly as the change in load increases. Minimum stiffness, reached during unloaded shortening, is independent of the initial force. As active muscles are lengthened, both peak and steady-state forces increase linearly, so that muscle stiffness remains constant and is likewise independent of the initial force. Remarkably, the steady-state stiffness of actively lengthening muscle approaches the minimum stiffness observed during active shortening at zero load. These observations support the hypothesis that titin plays a role in elastic recoil of actively shortening muscle as well as in steady-state enhancement of force with stretch. Supported by NSF IOS-0623791, IOS-0732949, NIH R25-GM56931, the TRIF Fund for Biotechnology and Science Foundation Arizona.
More boing for your buck: spring properties of muscle at and beyond optimal length
Leslie Gilmore, Jenna Monroy, Jim Hokanson, & Kiisa Nishikawa
A muscle produces maximal force when activated at its optimal length. If the muscle is rapidly unloaded at this length, it exhibits a fast recoil phase and then a slow phase of shortening. Distance and speed of recoil during the initial phase are determined by the elastic properties of the muscle, while the slow phase of shortening is a function of the cycling of the crossbridges with the thin filament. In this study, we investigated the effects of increases in muscle length on the fast recoil phase. We used a servo motor force lever system to perform load clamp experiments on mouse soleus and EDL muscles. Prior to activation, muscle lengths were increased 5-25% beyond optimal lengths in order to measure the distance of shortening during the fast phase. Preliminary results show that, for a given change in load, as muscle length increases the distance shortened during the fast phase is greater. Remarkably, the stiffness decreases as the muscle length increases beyond the optimal length. These preliminary results appear to be consistent with the hypothesis that the viscoelastic titin protein contributes to both force enhancement with stretch and elastic recoil during rapid unloading in active muscle. Supported by NSF IOS-0623791, IOS-0732949, NIH R25-GM56931, the TRIF Fund for Biotechnology and Science Foundation Arizona.
Don’t call me a stiff: changes in performance and histology with age
Morgan Burnette, Jenna Monroy, Bud Lindstedt, & Kiisa Nishikawa
As many of us can feel, muscles get stiffer and weaker as we age. Stiffness is dependent on the force generated by the muscle as well as the external load. This is true for muscles during both active shortening and lengthening. Our study investigates the histology underlying changes in stiffness and force during active shortening. Mouse soleus and EDL muscles were serially sectioned and stained for ATPase activity and oxidative capacity. We measured whole muscle cross-sectional area, fiber cross sectional area, fiber density, fiber-type (fast or slow), fiber-type ratio and distribution. Relative to old muscles, young muscles are larger in cross-sectional area and mass. In young EDL and soleus muscles, low oxidative fibers have greater cross-sectional area and diameter than high oxidative fibers. Furthermore, the ratio of fast to slow fibers was 60:40 in EDL and 50:50 in soleus. Preliminary results indicate that old EDL and soleus muscles have relatively fewer high oxidative fast fibers. As such, muscles that produce less force have increased stiffness. These results suggest an increased importance of maintaining muscle force to ensure more compliant muscles as we age.
It’s in the way that you use it: how activation patterns affect anuran feeding behavior
Eric Zepnewski, Carrie Carreno, Kris Lappin, & Kiisa Nishikawa
Most frogs and toads capture prey by protracting their tongues. More than 90% of the power for this movement is generated by a single pair of jaw opening (depressor) muscles. In species of Bufo, the depressor muscles are pre-loaded to achieve the relatively high forces necessary for ballistic tongue protraction. When the depressor muscles are pre-loaded, elastic energy is stored and then recovered during mouth opening. As such, the spring properties of the depressor muscles are thought to play an important role. In comparison, Ceratophrys cranwelli exhibits non-ballistic tongue protraction. Simultaneous EMG and high-speed video recordings show that the depressor muscles are not pre-loaded prior to tongue protraction. In situ muscle lever experiments in Ceratophrys demonstrated that active depressor muscle stiffness during shortening resembled stiffness values of Bufo when they were loaded similarly. Thus, behavioral differences may be explained by different morphologies. Morphological measurements showed differences in mechanical advantage between species. These results suggest that muscle is an adaptable tissue that, by virtue of common intrinsic elastic properties, may exhibit extremely different behaviors under varying conditions of activation, load, and mechanical advantage. Supported by NSF IOS-0623791, IOS-0732949, NIH R25-GM56931, the TRIF Fund for Biotechnology and Science Foundation Arizona.
Response of Herpetofauna to Ponderosa Pine Forest Treatments Prescribed by the National Fire and Fire Surrogate Study
Jean Block & Kiisa Nishikawa
Ponderosa pine forests across the southwestern United States are being treated by thinning and burning to reduce fuel loads and the risk of catastrophic wildfire and to reintroduce fire regimes that were known to occur prior to European settlement. However, little is known about how these treatments influence herpetofauna populations. Compared to bird and mammal studies, herpetofauna are underrepresented in the literature. Studying their response to forest treatments is especially important given their general decline worldwide. I compared post-treatment occupancy rates, abundance, sex and age ratios and survival of herpetofauna between four different forest treatments prescribed by the National Fire and Fire Surrogate Study in northern Arizona. Additionally, I examined associations between habitat and occupancy rate in each treatment. Treatments were prescribed in three replicated blocks and included an untreated control, a burned area, a thinned area and a thinned and burned area. Results from my study revealed several significant patterns. The southern plateau lizard (Sceloporus undulates tristichus) was the dominant species detected and was the only species that provided a sufficient sample size to perform statistical analyses. Occupancy rate of these lizards was significantly greater in the thin/burn treatment and across one replicate. The proportion of hatchlings was significantly lower in the untreated control. Habitat features associated with late successional forest stages were negatively correlated with occupancy while those associated with early successional forest stages were positively correlated with occupancy. My study suggests that southern plateau lizards select habitat associated with early to middle successional forest stages, especially habitat disturbed by the combination of thinning and burning. Additionally, untreated, dense, fire-suppressed forests restrict juvenile recruitment of this species. Increased presence of both adult and juvenile lizards in thinned and burned forest areas indicates increased diversity of other species. Hence, thinning and burning ponderosa pine forests to some degree is recommended. However, in the interest of other species, it is important to maintain patches of middle to late successional forest habitat. Finally, further studies of other herpetofauna species in treated and untreated ponderosa pine forests are recommended since very few of these species were detected.
The biomechanics of cephalopod branchial hearts: an interdisciplinary approach to a novel inspiration for the treatment of peripheral arterial disease
Ted Uyeno, Scott Nichols & Kiisa Nishikawa
Peripheral Arterial Disease concerns Arizona because of its prevalence and debilitating symptoms. Caused by reduced blood flow through peripheral arteries, surgical treatment may involve implanting synthetic vascular grafts. Because these rigid implants tend to buckle and plug with clots, new designs are of great value. Our goal is to develop novel design principles using a biologically inspired approach: as active cephalopods evolved efficient, closed-circuit, and high-pressure circulatory systems with elastic arteries and accessory hearts, from open and low-pressure ancestral precursors, biomechanical analyses may inspire useful structural designs. To identify principles of function, we will describe cephalopod accessory heart morphology using microscopy, histology, and corrosion casts; we will test vascular wall mechanical properties using force lever and material properties testing; and electromyography, cinematography, and pressure measurements will be used to test the resultant functional postulates. These data will enhance future grant proposals to develop proof of concepts for novel implant designs.